Iron and Zinc Interactions
As compared to the amount of necessary organic nutrients, the amount of inorganic nutrients required for human and animal life is remarkable. A large number of purposes are used by certain inorganic materials, such as iron and zinc, while others, such as selenium and molybdenum, have only a few applications. The variations are relatively great in the identities of the different inorganic nutrients.
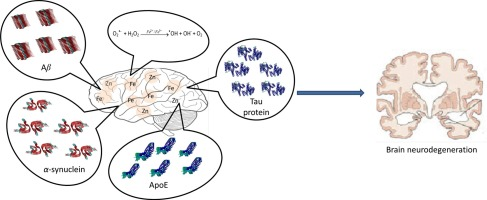
The fact that copper deficient rats can experience iron deficiency anaemia, as revealed by haemoglobin and hematocrit measurements, indicates a relationship between copper and iron. In a special iron transport process, ceruloplasmin can also act. Iron is low-cost, non-toxic and very available. 1538oC is the melting point of iron. In the Earth’s surface, iron is the most common transition metal. Thus, iron has long been the most popular metal in our daily life and industry.
Zinc Uses in Everyday Life
Long before zinc was known as a metal. Romans had learnt to produce brass, an alloy of copper and zinc, by smelting calamine with copper ores. It is the last element of the first transition series and the first member of group 12. Zinc is used by a great number of enzymes and proteins, whereas copper seems to be limited to only a few functions. The zinc uses are listed below.
-
as an alloying element in aluminium and copper alloys
-
for galvanizing steel and
-
for die castings.
The principal die casting alloy is aluminium and magnesium. It is used widely for parts of locks and other small parts, handles, carburetor and fuel pump housings. Die casting accounts for approximately 1/3 of the annual consumption of zinc. Of the three metals used for most die castings, zinc, magnesium and aluminium, zinc has the lowest melting point. Wear of the dies is least, so zinc die castings usually have the best surface finish. However, because of its relatively high density, zinc is being replaced by lighter metals and plastics in applications where weight is of concern.
Malleable and Ductile Properties of Iron
Ductile cast iron has about the same compositions as grey iron except that the sulfur content is very low. Magnesium is added to the melt just before pouring to further reduce the sulfur. Heating a white cast iron to just below the eutectic temperature allows the iron carbide to decompose. The graphite forms as clumps or rosettes called temper carbon. The properties are similar to ductile cast iron. The heat treatment required to form malleable iron is expensive.
The two categories of iron depend on the percentage of impurity. Cast iron contains 4% carbon and small portions of silicon, phosphorus and sulphur as impurities. These impurities make it hard and brittle. It is used for making furnaces, grates, railing, engine blocks, iron boxes etc. Wrought iron contains 0.1% carbon. It is malleable and can easily be forged. It is used to make iron nails, iron sheets, horseshoes, agricultural implements etc. Cast iron is used to make drain pipes, lamp posts etc. Wrought iron is used to make chains, ornamental gates etc. The most important use of iron is in making alloys like steel and stainless steel.


